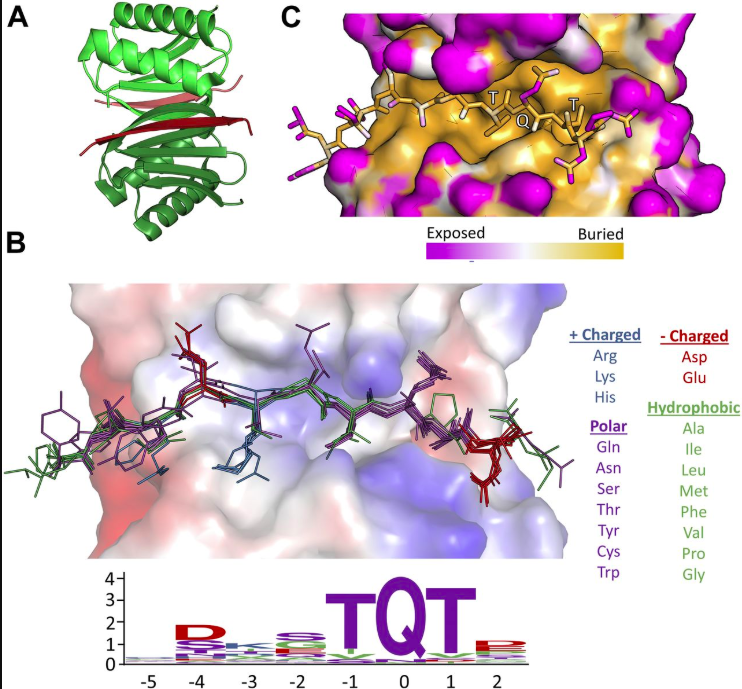
Most proteins in interaction networks have a small number of partners, while a few, called hubs, participate in a large number of interactions and play a central role in cell homeostasis. One highly conserved hub is the eukaryotic protein LC8, which was originally identified as an essential component of the multi-subunit dynein complex, is essential in multiple protein complexes in diverse systems. Indeed, LC8 is broadly distributed troughout the cell, and its more than 100 verified partners can be found in many cellular compartments such as the nucleus, mitochondira, and vesicles. Intriguingly, LC8 does not passively bind its various partners, but emerging evidence instead suggests that LC8 acts as a dimerization engine that promotes self-association and/or higher order organization of its primarily disordered, monomeric partners. This structural organization process is triggered by long-range allosteric regulation initiated by LC8 binding a pair of disordered chains, forming a bivalent or polybivalent scaffold.
To learn more about hub protein, LC8, you can visit our website: LC8 Hub and to find out if your protein binds LC8, you can navigate to LC8PRED and use our algorithm developed to identify LC8 binding partners. To learn even more, see our publication, "Systematic identification of recognition motifs for the hub protein LC8".